 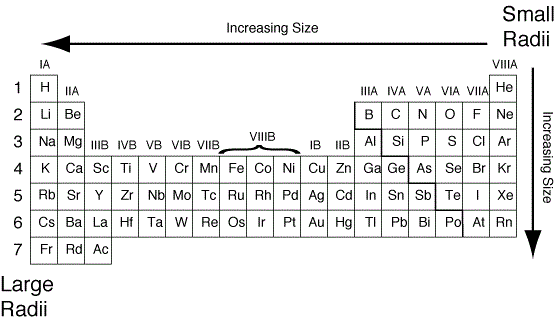
It is often denoted by a 0 and is approximately 53 pm. The Bohr radius is consequently known as the "atomic unit of length". Just as atomic units are given in terms of the atomic mass unit (approximately the proton mass), the physically appropriate unit of length here is the Bohr radius, which is the radius of a hydrogen atom. For more recent data on covalent radii see Covalent radius. Note: All measurements given are in picometers (pm). These trends of the atomic radii (and of various other chemical and physical properties of the elements) can be explained by the electron shell theory of the atom they provided important evidence for the development and confirmation of quantum theory. The radius increases sharply between the noble gas at the end of each period and the alkali metal at the beginning of the next period. For instance, the radii generally decrease rightward along each period (row) of the table, from the alkali metals to the noble gases and increase down each group (column). Ītomic radii vary in a predictable and explicable manner across the periodic table. Under some definitions, the value of the radius may depend on the atom's state and context. 
Depending on the definition, the term may apply only to isolated atoms, or also to atoms in condensed matter, covalently bound in molecules, or in ionized and excited states and its value may be obtained through experimental measurements, or computed from theoretical models. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. An A-Z Guide to the Elements.The atomic radius of a chemical element is the distance from the center of the nucleus to the outermost shell of an electron. We wish we had saved one of the first edition videos, for the sake of comparison. Now the videos stream and the CD seems no longer on offer. 
The animations in the original were huge for the time (hydrogen was about 12 megabytes), so downloading and saving them was a wise use of bandwidth-or one could buy a CD for $37 (20 pounds). The current version is at least the second. Society of Chemistry (UK) are truly exceptional. resourcesīoth the content and design of this site created by the Royal Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides.Īcta Crystallographica, Section A. The coordination properties and ionic radius of actinium: A 120-year-old enigma.Ĭoordination Chemistry Reviews, vol. Gauthier J.-P.Deblonde, Mavrik Zavarin, and Annie B. From this pattern scientist are able to distance ebetween the atoms and from thst, Elementġ. I one shines x-rays through the crystel onto a sensor, such as a sheet of photographic film, a pattern of spots. The atoms in a crystel are arannged in a regular pattern. They are cfound in clouds.ĭistance beteen teh central charge and the outermost electron. n atom is not like a minature solre sysem either, though it is sometimes helpful to think of it that wat.An atoms electrons don't travil in regualr orbits. We know it has a posoitively charged center (the nucleus), which is surounded by mving electrosn. 
What does it mean to measure the “size” of an atom?Īn atom is not like a billiard ball. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
